【Organic Chemistry】【Takashi OOI】A researcher’s uniqueness—what have I accomplished?

Takashi OOI (Ph.D.)
Professor, Institute of Transformative Bio-Molecules, Graduate School of Engineering, Nagoya University
—
Prof. Ooi’s research, centered on organic ion pair catalysis, is unique in the world. Currently conducting research that combines radical reactions as a KAKENHI* recipient project in the category of Transformative Research Areas (A) Green Catalysis Science for Renovating Transformation of Carbon-Based Resources, he continues to communicate new findings in Chemistry to the world.
*The Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research
—
As an undergraduate student, Prof. Ooi wondered whether he should go on to a doctoral course. Until then, he had been working hard, carrying out experiments day and night and gaining some confidence as a young researcher. Yet, his critical mind had some doubts: The world is vast and filled with diverse people. So would he want to spend the rest of his life in the lab?
He considered the option of a corporate job. As he imagined himself lined up with other students from various universities in an interview room, he wondered why he should be selected and whether he represented something special that would justify a salary. His own answer was, “Not really.”
Although he had achieved some research results by then, he himself had never come up with research ideas from scratch. He thought that he had simply been working hard. In such a situation, his mentor, then Associate Prof. Keiji Maruoka (now Designated Professor, Graduate School of Pharmaceutical Sciences, Kyoto University), suggested that he enter a doctoral program. He felt encouraged, noticing that someone believed, even if only a little, that he should pursue that path.
He imagined himself doing his own research with his own ideas, which he would test in experiments, exploring unknown spheres of Chemistry. He thought about the kind of scientist he wanted to be, someone who would accomplish something unique through teamwork with those with whom he would build a relationship of trust. Yes, I’m going to do a Ph.D., he decided. Since then, he has been asking himself, “Is there something unique only I can do?”

Looking back on his journey so far, Prof. Ooi says: “To put it a little dramatically, I think there’s real value in trying something and failing. All the ideas accumulated through each research project that never made it into papers will eventually lead to something, and this is what makes the uniqueness of each researcher.”
What appears in research papers is just the tip of the iceberg. A researcher’s treasure lies in the vast number of experiments and numerous negative data that are behind published results. Prof. Ooi says that relying on efficacy and good ideas is correct in some ways, but he also believes that research results coming solely from them will not stand the test of time.
Prof. Ooi’s experiment-filled life began in his fourth undergraduate year, when he was assigned to a research lab.
Support from mentors during his experiment-filled days
He remembers being “a lazy student.” Prof. Hisashi Yamamoto (now Professor Emeritus at Nagoya University) and Prof. Maruoka taught him organic chemistry from scratch and mentored him all along. They played such a major role in his career that he says, “My meeting with them was the starting point for everything.” When he was a fourth-year undergraduate, a paper he co-authored with them was published in the prestigious American Chemical Society’s journal. It is a memorable paper for him, with his name appearing between his two mentors’ names.

Compounds, although made of the same material, can be made completely different if shaped differently, even only slightly. As an example, let us take a look at some steps of the experiment that the three researchers conducted in the process of producing results that led to the paper mentioned above. Do you notice in Fig. ① and ② the difference in the shape of the starting materials and in the shape and number of the resulting compounds?

In both ① and ②, the starting material contains 12 carbon atoms (C), 24 hydrogen atoms (H), and one oxygen atom (O), but their configurations differ slightly. Aluminum (Al) in the catalyst coordinates with O. In ①, it is unclear which of the two carbon bonds will break, but in ②, only the bond on the left carbon is selectively broken.
In the very early stage of the experiment, a jumble of many different compounds with seemingly similar yet essentially different structures came about. Then, suddenly one day, it became possible to selectively obtain just one compound. He did not immediately understand how this worked. He was simply surprised by the results and thought, “Something’s different.”

At first, Prof. Yamamoto advised a young Mr. Ooi to change his attitude. Prof. Maruoka often gave him an earful as they spent all their time in the lab. Gradually, however, he began to find experiments more interesting. Preparation for the graduate school entrance exam was the first time he studied seriously since entering university. His hard work to stay in the lab as a graduate student resulted in excellent grades. Prof. Maruoka was pleasantly surprised. Prof. Yamamoto also sensed the change in Mr. Ooi. When he was in his second year of graduate school, Prof. Yamamoto personally assigned him to tutoring some younger students. He still treasures the moment he was given this task.
“For Prof. Yamamoto, it may have been nothing special, but it made me very happy,” says Prof. Ooi.

Capturing opportunities while working with lab members
Prof. Ooi continued to work on many research projects with Prof. Maruoka, who provided him with the opportunity to work as an assistant professor at Hokkaido University and an associate professor at Kyoto University. They also worked together in opening new laboratories and exploring new research topics of chemistry. By that time, Prof. Maruoka had come to treat his mentee as a fellow researcher.
“I was fortunate to be able to work while imagining what Prof. Maruoka was thinking in real-life situations as a scientist,” says Prof. Ooi.
When Prof. Ooi opened his own laboratory at Nagoya University, he started with absolutely nothing but was at ease. His policy was to take advantage of the opportunity he was given and simply do what he wanted to do. He was joined by Dr. Daisuke Uraguchi (then assistant professor, now Professor at Hokkaido University Institute for Catalysis) and five students who chose the laboratory as their first choice. Having such good members was the best driving force for Prof. Ooi and the laboratory.
One day, Prof. Ooi decided to start “something new,” and he began working on catalytic chemistry of organic ion pairs. One of the lab’s research achievements, which fully utilized Prof. Uraguchi’s area of expertise, was aminophosphonium salt. To briefly explain just a few of the characteristics of this catalyst, it uses the attractive forces between positive and negative charges, the power of hydrogen bonds, and the three-dimensional structure of the catalyst to capture and control starting-material molecules, realizing the selective chemical synthesis of products with a desired structure.
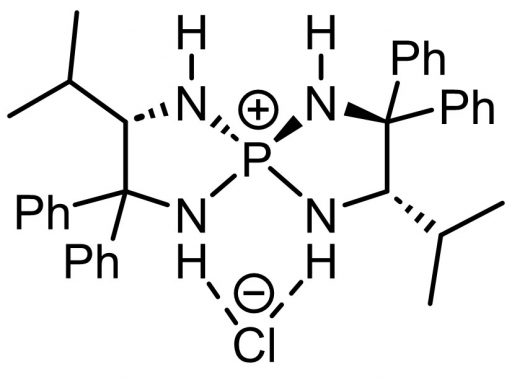
(Top) A form of aminophosphonium salt, which takes on various shapes; positively charged catalyst molecule attracts negatively charged molecules and captures them at the site of hydrogen bonding. In this form, a Cl– (chloride ion) is attached.
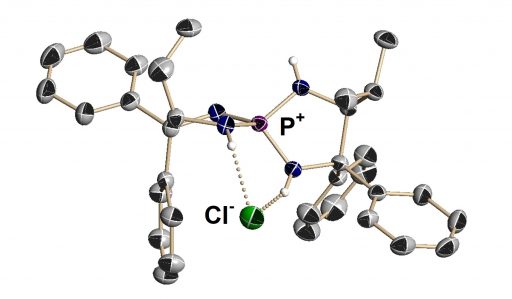
(Middle) A three-dimensional structure determined by X-ray structural analysis; it has a compact structure, but it wraps around and fixes the molecule.
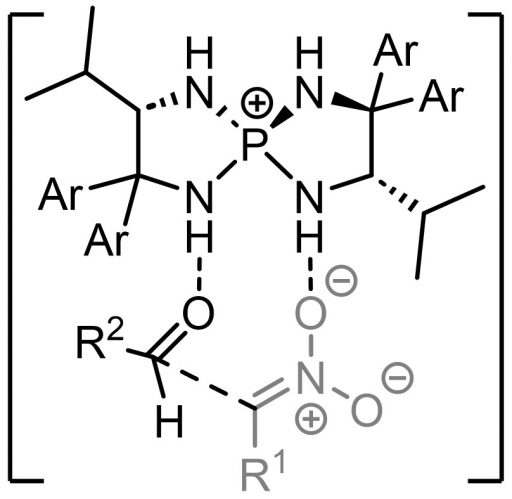
(Bottom) An example of how a catalyst works; reaction is controlled where Cl– was attached. For greater detail, refer to the paper below:
Chiral Tetraaminophosphonium Salt-Mediated Asymmetric Direct Henry Reaction; Daisuke Uraguchi, Sawako Sakaki, and Takashi Ooi; J. Am. Chem. Soc. 2007, 129, 12392-12393.
This catalyst was further developed thanks to great efforts by the lab’s student. He explored the possibility of replacing the Cl– site with another interesting molecule and, starting from scratch, chose phenol, which is readily available in a wide variety of commercial forms.
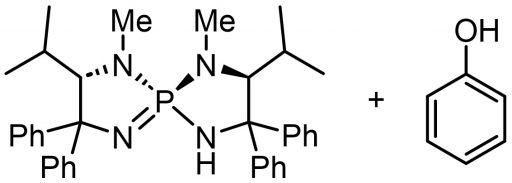
At first, they imagined a structure in which one aminophosphonium was attached to one phenol. The synthesis reaction went reasonably well. However, they were not able to explain why replacement with phenol was necessary. Then one day, the student in charge of this experiment came up with the idea of trying X-ray structural analysis, which revealed a surprising molecular structure. It turned out that there was not just one phenol but two more in between. This gave birth to new three-dimensional structures and made it possible to add various other ingenuities, such as attaching Cl to the phenol.
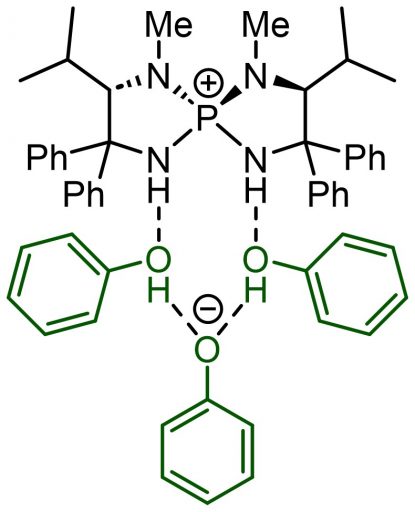
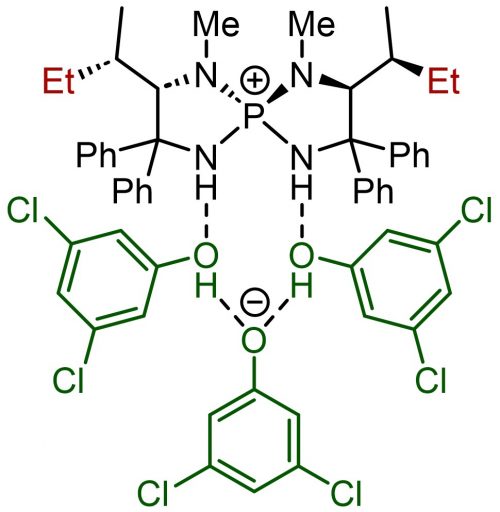
(Top) Aminophosphonium with three phenol molecules attached
(Bottom) Rearrangements with substituents on aminophosphonium and phenol molecules enabling further improvement on catalyst
The scientists often made a breakthrough by accident. It was serendipity in action. Their paper was published in Science, where the student’s name appeared between the names of his two supervisors. The story of young Mr. Ooi was repeated.

Encounters with people and repeated experiments leading to new chemistry
When Prof. Ooi takes on a new challenge, there is no hesitation. Even if successful outcomes seem unlikely, he forges ahead with his teammates on a project, as long as it has inherent originality.
“It’s not about whether there’s hope ahead. It’s about whether you do it or not. In the dark, taking a step forward brings about a ray of hope.”
Prof. Ooi has been supported by many people and given opportunities. Building on the foundation of numerous experiments he has conducted with the lab members, he simply continues in pursuit of something unique that only he can do.
“I am hoping to do the kind of research in chemistry that makes people around the world think and say, ‘Isn’t this from Ooi’s lab?’ even if the findings are published without the authors’ names or university name.”
(Interview and Text: Tatsuro AYATSUKA, Interview Date: September 17, 2025)
